My Green Life 4 R's
(reduce, reuse, repurpose, recycle)
Sewer Science ~ Water Recycling
Waste Water Treatments
The goal of Waste Water Treatment is to make sewage safe to be put in the ocean and clean for life in the water. However, it is not safe to drink.
Four tests
pH - pH indicates
how acidic or basic a solution is. The pH scale ranges from 0 to 14. Lower pH
means that the solution is acidic; higher pH means that the solution is basic.
The neutral pH is 7, and it is preferable for most life. Fish, shellfish,
birds, and plant life need the pH to be fairly neutral to survive. In addition,
bacteria at the wastewater treatment plant are most effective at eating the
pollution if the pH is between 6.6 and 8.5. The pH of the wastewater needs to
be tested to see if the pH is within that range. If it is not, we need to add a
chemical to change the pH. pH strips are used to test pH.
Ammonia - Ammonia (NH3) is highly toxic and is found in urine. Urine contains urea (ammonia and carbon dioxide). The bacteria naturally existing in a body of water will "attack" the ammonia in a way that "steals" precious amounts of dissolved oxygen. The oxygen that is used is necessary for the survival of species in water. Therefore, ammonia needs to be removed from wastewater. It is removed by bacteria (Secondary Treatment). Ammonia is measured in milligrams/liter (mg/L). Hach Ammonia Test Strips are used to measure ammonia concentration.
Turbidity - Turbidity
measures the concentration of suspended particles, or solid matter of various
sizes, in a solution. It measures clarity of the sample, not the color. Turbidity
is important because fine particles, silt, clay, and organic and inorganic
matter can interfere with oxygen absorption in fish’s gills. Also, as
wastewater moves through a treatment system, it becomes more and more clear. Turbidity
can be an indicator of wastewater treatment progress. When light passes through
the water, the particles suspended in the water causes the light to scatter; a
colorimeter, or spectrophotometer, measures turbidity using this property. The
turbidity of clean pure water is zero, but zero turbidity does not guarantee
that the sample is pure water.
COD - Chemical Oxygen Demand (COD) indicates the amount of oxygen that that would be needed to destroy all the pollution in the water. When the COD is higher, the water is more polluted. Dissolved oxygen reacts with pollution in the water and breaks it down into simple compounds. If the COD is high, then the pollution will be oxidized and the amount of oxygen available to marine life will be reduced. For example, when wastewater is dumped into a bay, the pollution is slowly altered or destroyed by bacteria and chemicals in the water. This process requires oxygen, so fish have less oxygen to survive.
Treatments

Raw Sewage
5/10/12
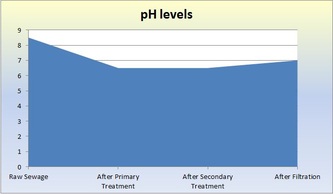
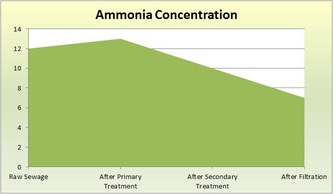
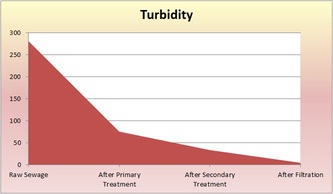
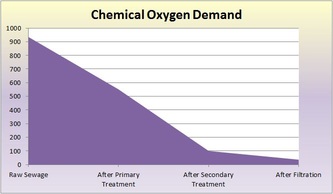
Raw - This is raw sewer water that has not been treated. It still contains both large and small particles of things not water. The solution we used to represent sewage consisted of 8g of dried used coffee grounds, 4.52g of torn-up toilet paper pieces, 3.44g of ground-up breakfast cereal, 3.94g of ground-up pet food, 4g of baking soda, 20mL of vegetable oil, 1.7g of cut-up plastic, and 10mL of ammonia. Normally in sewage, there would be paper products, like toilet paper, cleaning products, human waste, oils, and food. The pH of the raw solution was 8.5, the ammonia concentration was 12mg/L, the turbidity was 281, and the COD (chemical oxygen demand) was 935mg/L.

After Primary Treatment
5/14/12
Primary Treatment - This first treatment removes large solids and oil scum. It typically decreases turbidity and COD. Sedimentation and flotation occurs. As the sewage sits in a tank for 24 hours, the particles settle and separate by density: more dense sediment sinks to the bottom and less dense particles float to the top. Dissolved matter with the same density as water stays in the middle layer. The pH of the solution after sedimentation was 6.5, the ammonia concentration was 13mg/L, the turbidity was 75, and the COD was 551mg/L.

After Secondary Treatment
5/16/12
Secondary (Biological) Treatment - The middle layer of dissolved matter is poured into an Aeration Tank. Microorganisms eat the dissolved ammonia & food nutrients in the wastewater. Air is added for the aerobic microorganisms. Microorganisms eat the pollutants and convert the compounds into solids; it can later be separated from the water. As stated above, ammonia is toxic and must be removed from the wastewater; bacteria are able to convert ammonia into nitrate (nitrification).
The wastewater is transferred to the Secondary Sedimentation Tank so flocculation can occur (microbes settle to the bottom of the tank). Aluminum Sulfate ("alum") is added to capture the microorganisms and make them float to the bottom. The addition of alum often decreases the pH of the water. The pH of the solution after the biological treatment and flocculation was 6.5, the ammonia concentration was 10mg/L, the turbidity was 33, and the COD was 100mg/L.
The wastewater is transferred to the Secondary Sedimentation Tank so flocculation can occur (microbes settle to the bottom of the tank). Aluminum Sulfate ("alum") is added to capture the microorganisms and make them float to the bottom. The addition of alum often decreases the pH of the water. The pH of the solution after the biological treatment and flocculation was 6.5, the ammonia concentration was 10mg/L, the turbidity was 33, and the COD was 100mg/L.

After Filtration
5/16/12
Final/Tertiary (Filtration) Treatment - Filtration is our last treatment. Wastewater is poured through a filtration column of charcoal and sand. The filter removes very fine particles that are not dense enough to sink to the bottom and are not eaten by bacteria or other microbes. The pH of the solution after filtration was 7, the ammonia concentration was 7mg/L, the turbidity was 4, the COD was 36mg/L.
Sometimes, disinfection is the last stage of water treatment. Ultraviolet radiation or chlorination is used to destroy organisms that may endanger human health.
Sometimes, disinfection is the last stage of water treatment. Ultraviolet radiation or chlorination is used to destroy organisms that may endanger human health.
Summary Data Table
|
Tests
pH
Ammonia concentration (mg/L)
Turbidity
Chemical Oxygen Demand (mg/L)
|
Raw
5/10/12 8.5
12
281
935
|
After Primary Treatment 5/14/12
6.5
13
75
551
|
After Secondary Treatment 5/16/12
6.5
10
33
100
|
After Filtration
5/16/12 7
7
4
35
|
Summary Graphs
Summary Analysis
jgbvtycrtujc vkjk mowow poicfgvbh water gvhjlkbyufvihbjnm hj Not all gibberish. The wastewater treatments cleaned sewage and made it safe to be put in the ocean. It is not safe to drink. The ammonia did not decrease until after the secondary treatment because the bacteria from the secondary treatment removed ammonia from the wastewater. It is good that the ammonia levels decreased because ammonia is extremely toxic. The turbidity decreased throughout the treatments, but it decreased the most after the primary treatment. This makes sense because the sewer water had large particles and chunks of junk. After the primary treatment, those large particles were removed; they either floated or sunk. This made the water much more clear than than the raw sewage. During the next treatments, the water continued to become more clear, which resulted in lower turbidity levels. Supposedly, the addition of alum during the Secondary treatment lowers pH. This was not apparent in our results. However, the pH did end up as 7, which is very good for the results of the wastewater treatments and marine life in general. The COD decreased drastically up until the secondary treatment. After filtration, it lowered slightly. Because COD represents pollution in the water, the results make sense. Most of the pollution would be removed from the water sample because the primary treatment got rid of large solids and oil scum, and the secondary treatment got rid of other dissolved matter. Therefore, the water would contain considerably less pollutants after those treatments. Filtration removes the last bits of pollutants, so the COD did not decrease as much. In conclusion, the water waste treatments significantly cleansed the wastewater. It became much more safe and would not endanger marine life.
Reducing Your Water Footprint
I took the Water Footprint Quiz, and was a little bit surprised. The US average for water used in the home is 100; my home was 109. The average for water used for our diet was 1,056; my diet was 977. Water used for transportation and energy was 700; my transportation was 362. Average water used by stuff was 125; my stuff was 40. I take a lot of flights so I was surprised that I had such a low amount of water use for transportation and energy compared to the US average. I suppose it is because I do not drive, and do not continuously use energy for transportation in that way. I have a lower than average water usage for diet because we do not eat as much meat as others. I also have lower than average water usage for stuff simply because I do not get too many new things. However, I have a higher than average water usage for our house. We use quite a bit of water for everyday things such as showers, washing dishes, and washing clothes. This is related to the My Green Life Project because it has to do with water usage and allows people to be more aware of the effect of what they do on the environment.